The rapid increase of CO2 content in the atmosphere caused serious greenhouse effect and some other environmental problems. The capture, storage, conversion and utilization of CO2 (CCUS) is an efficient way to solve these issues. Among them, CO2 hydrogenation to hydrocarbons can not only reduce the CO2 emission, but also open up a new route for the production of value-added chemicals, as well as effectively alleviate the energy crisis caused by the reduction of oil resources.Nonetheless, it is very challenging to achieve a high selectivity to a specific hydrocarbon (in particular a relatively long-chain one) through CO2 hydrogenation, due to the chemical inertia of CO2 and the complexity of C-C coupling. Therefore, the key of CO2 hydrogenation to hydrocarbons is to control the reaction pathway for hydrocarbons formation. There are two routes for CO2 hydrogenation to hydrocarbons. One is the modified Fischer-Tropsch (FT) synthesis route, which shows a high CO2 conversion. However, due to the restriction of Anderson?Schultz?Flory (ASF) rule, it is difficult to controllably control the distribution of hydrocarbon products, and the selectivity of methane can reach 15-25%. In contrast, conversion of CO2 to hydrocarbons through the “methanol-intermediate route” has more potential, because it effectively breaks the restrictions of ASF rule.
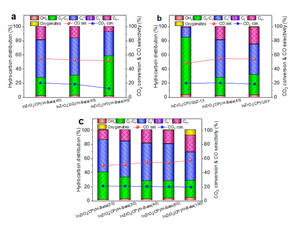
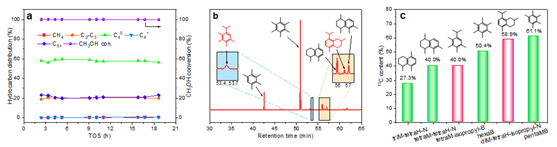
Despite important advances, achieving high selectivity for the production of single or specific hydrocarbon from CO2 hydrogenation remains a challenge. Recently, the research team designed a InZrOx/H-Beta bifunctional catalyst, which showed high butane selectivity (~53.4%) in CO2 hydrogenation (Figure 1). Combined with the results of 12C/13C methanol switching experiment and DFT calculations, it is confirmed that the three-dimensional 12-MR channel of H-Beta is conducive to the formation of intermediates such as methylbenzene and methylnaphthalene species with isopropyl side chains, and butane can be easily formed through rapid alkyl elimination, methylation and hydrogenation reactions (Figure 2).
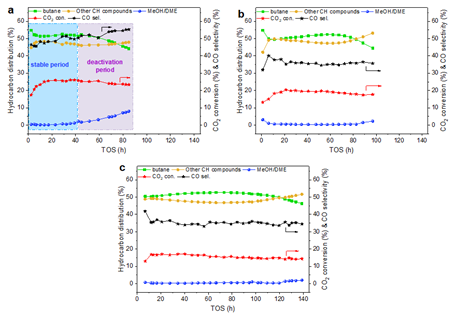
In addition, they proposed the surface silica protection strategy to enhance the interaction between SiO2 and surface indium species, which can obviously inhibit the phase separation of InZrOx oxide and the indium migration, and thus, considerably improving the catalytic stability of InZrOx/H-Beta in the hydrogenation of CO2 to hydrocarbons (Figure 3). Figure 1. Catalytic performance of InZrOx/H-Beta composite catalysts in CO2 hydrogenation to butane.
Figure 2. Methanol conversion probe experiment in H-Beta zeolite.
Figure 3. Catalytic performance of silica modified InZrOx/H-Beta composite catalysts in CO2 hydrogenation to butane.
This work was published in Nature Communications (2023, 14, 2627). It was funded and supported by the National Natural Science Foundation, National Key R&D Program, the Shanxi Provincial Excellent Youth Project, the Youth Innovation Promotion Association of the Chinese Academy of Sciences and the National Supercomputing Center of Lvliang.