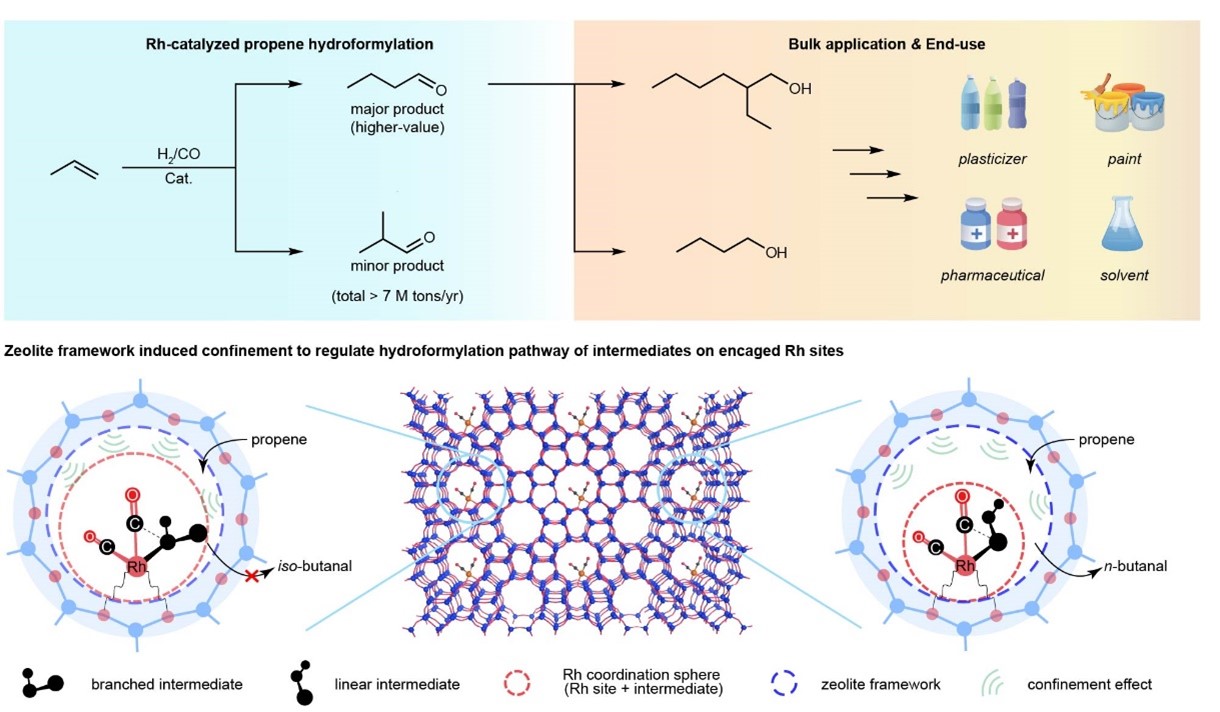
The rhodium (Rh)-catalyzed hydroformylation of propene to produce butanal represents one of the foremost homogeneous catalytic processes in the chemical industry. The indispensability of butanal in the manufacture of a multitude of daily essential products (e.g., plasticizers, detergents, paints and pharmaceuticals) underscores the significance of this industrial process. While the process has achieved a commendable level of maturity and efficiency, the enduring challenge of Rh metal loss (notably during catalyst separation and recycling) remains, as it intricately influences the economic viability of the technology. This concern is further accentuated by the remarkable surge in Rh prices with nearly 50-fold from the 1970s to the 2020s. This substantial escalation in costs assumes particular prominence when considering the global capacity for propene hydroformylation, which exceeds 7 million tons per year. Therefore, tremendous efforts have been devoted to develop supported hydroformylation catalysts, which usually take advantage in facile catalyst separation and recycling, to the tackle the above problem. However, supported Rh centers consist of rotationally flexible and sterically unconstrained Rh hydride dicarbonyl centers which lead to limited regioselectivities and largely impeded the practical implementation of supported hydroformylation Rh catalysts.
Recently, a groundbreaking achievement was made by a collaborative team from Institute of Coal Chemistry, Chinese Academy of Sciences, andSynfuels China Technology Co., Ltd. They developed a novel supported "Rh-zeolite" catalytic system and achieved unprecedented regioselectivity in propene hydroformylation for the production of n-butanal. The team extended the classical shape-selectivity concept within zeolite catalysis to the zeolite confined Rh sites, which enables to steer the reaction intermediate toward formation of n-butanal. The optimized catalyst achieved over 99% regioselectivity for the desired product n-butanal, with total aldehyde selectivity also exceeding 99%, and a catalyst turnover frequency exceeding 6500 h-1, surpassing all the supported catalysts and nearly all homogeneous catalysts developed so far.
This research not only provides a novel method for selectively production of thermodynamically less stable (anti-Markovnikov) n-butanal via supported catalysts but also extends the concept of shape-selectivity offered zeotypes to the extra-framework metal sites. It also offers new perspectives and methodologies for addressing the significant issues circulated in industrial propene hydroformylation processes.
On April 24, the research findings were published in Nature. The study received support from the National Key R&D Program, the National Natural Science Foundation of China, and research funding from Synfuels China Technology Co., Ltd.
Industrial propene hydroformylation and proposed supported Rh-zeolite system.