Fiber reinforced thermosetting resin composites, especially carbon fiber reinforced epoxy resin composites and glass fiber reinforced unsaturated polyester resin composites, are widely applied in aviation, spaceflight, automobile, shipping, circuit board, wind power, etc. The recycling methods of thermosetting resin mainly include mechanical, thermal and chemical recycling means. And the chemical recycling is the most promising recycling strategy. Currently, chemical recycling process mainly use high temperature or strong oxidants to destroy the chemical structure of resin matrix in order to get the degradation products and fibers, which is nagged by disadvantages such as complex degradation products, poor cycle utilization and heavy damage of the fibers.
Research Group 311 of ICC put forward a novel idea of selective cleaving chemical bond degradation to recover thermosetting resin. By using incompletely coordinated zinc ions or weakly coordinated aluminum ions to selectively cleave chemical bond in resins, the efficient degradation and completely recycling of thermosetting resin-based composites was realized. And by using incompletely coordinated zinc ions in aqueous solution to selectively cleave the carbon-nitrogen bond of epoxy resin, the efficient degradation and recycling of carbon fiber reinforced epoxy resin composites was realized as well. (Green Chem., 2015, 17, 2141-2145,http://pubs.rsc.org/en/Content/ArticleLanding/2015/GC/C4GC02512A#!divAbstract). Furthermore, the recycling of glass fiber reinforced unsaturated polyester resin composites was realized via using weakly coordinated aluminum ions in acetic acid solution to selectively break the ester bond. (Green Chem., 2015, 17, 4527-4532,http://pubs.rsc.org/en/Content/ArticleLanding/2015/GC/C5GC01048A#!divAbstract). Three national invention patents have been applied, and one of them has been authorized (ZL 201310163799.1,201410562107.5,201510056492.0).
The recycling of thermosetting resins is very difficult because of their insoluble and infusible properties incurred by their three-dimensional structure. Presently, economical and green opening of the stable C-N and C-O bonds of chemicals is an research hotspot in catalysis and environmental field. The challenge of selective bond cleavage is the selective activation of the active sites of reactant. Incompletely coordinated zinc ions and weakly coordinated aluminum ions have intense complexation tendency, and are readily to interact with the heteroatom, therefore weakening the carbon heteroatom bonds and causing its cleavage. The recycling process developed by ICC only causes little damage on recycled fibers, and more importantly, the degradation products are composed of chemicals with clear structure and can be easily recycled. Research on the selective cleavage of C-N and C-O bonds by controlling the coordination state of metal ions is of great scientific and practical significance.
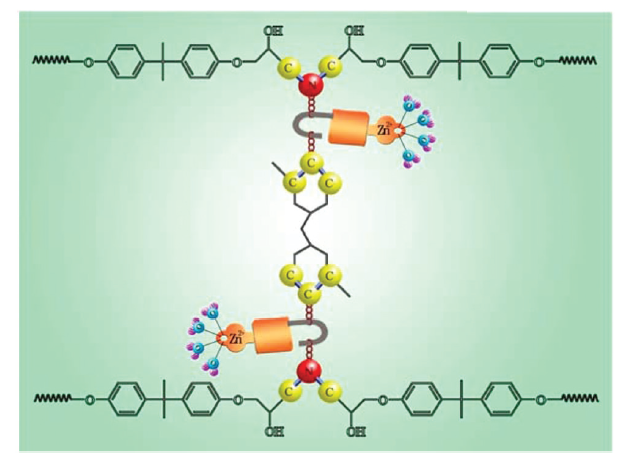
Figure 1. Selective cleavage of C-N bonds to recycle CF/EP composites
Figure 2. Selective cleavage of ester bond to recycle GF/UPR composites
